Mark Dominus (陶敏修)
mjd@pobox.com

Archive:
| 2026: | JFM |
| 2025: | JFMAMJ |
| JASOND | |
| 2024: | JFMAMJ |
| JASOND | |
| 2023: | JFMAMJ |
| JASOND | |
| 2022: | JFMAMJ |
| JASOND | |
| 2021: | JFMAMJ |
| JASOND | |
| 2020: | JFMAMJ |
| JASOND | |
| 2019: | JFMAMJ |
| JASOND | |
| 2018: | JFMAMJ |
| JASOND | |
| 2017: | JFMAMJ |
| JASOND | |
| 2016: | JFMAMJ |
| JASOND | |
| 2015: | JFMAMJ |
| JASOND | |
| 2014: | JFMAMJ |
| JASOND | |
| 2013: | JFMAMJ |
| JASOND | |
| 2012: | JFMAMJ |
| JASOND | |
| 2011: | JFMAMJ |
| JASOND | |
| 2010: | JFMAMJ |
| JASOND | |
| 2009: | JFMAMJ |
| JASOND | |
| 2008: | JFMAMJ |
| JASOND | |
| 2007: | JFMAMJ |
| JASOND | |
| 2006: | JFMAMJ |
| JASOND | |
| 2005: | OND |
Subtopics:
| Mathematics | 246 |
| Programming | 100 |
| Language | 95 |
| Miscellaneous | 75 |
| Book | 50 |
| Tech | 49 |
| Etymology | 36 |
| Haskell | 33 |
| Oops | 30 |
| Unix | 27 |
| Cosmic Call | 25 |
| Math SE | 25 |
| Law | 23 |
| Physics | 21 |
| Perl | 17 |
| Biology | 16 |
| Brain | 15 |
| Calendar | 15 |
| Food | 15 |
Comments disabled
Sat, 13 Jan 2018
How do plutonium-powered pacemakers work?
I woke up in the middle of the night wondering: Some people have implanted medical devices, such as pacemakers, that are plutonium-powered. How the hell does that work? The plutonium gets hot, but what then? You need electricity. Surely there is not a tiny turbine generator in there!
There is not, and the answer turns out to be really interesting, and to involve a bunch of physics I didn't know.
If one end of a wire is hotter than the other, electrons tend to diffuse from the hot end to the cold end; the amount of diffusion depends on the material and the temperature. Take two wires of different metals and join them into a loop. (This is called a thermocouple.) Make one of the joints hotter than the other. Electrons will diffuse from the hot joint to the cold joint. If there were only one metal, this would not be very interesting. But the electrons diffuse better through one wire (say wire A) than through the other (B), and this means that there will be net electron flow from the hot side down through wire A and then back up through B, creating an electric current. This is called the Seebeck effect. The potential difference between the joints is proportional to the temperature difference, on the order of a few hundred microvolts per kelvin. Because of this simple proportionality, the main use of the thermocouple is to measure the temperature difference by measuring the voltage or current induced in the wire. But if you don't need a lot of power, the thermocouple can be used as a current source.
In practice they don't use a single loop, but rather a long loop of alternating metals, with many junctions:
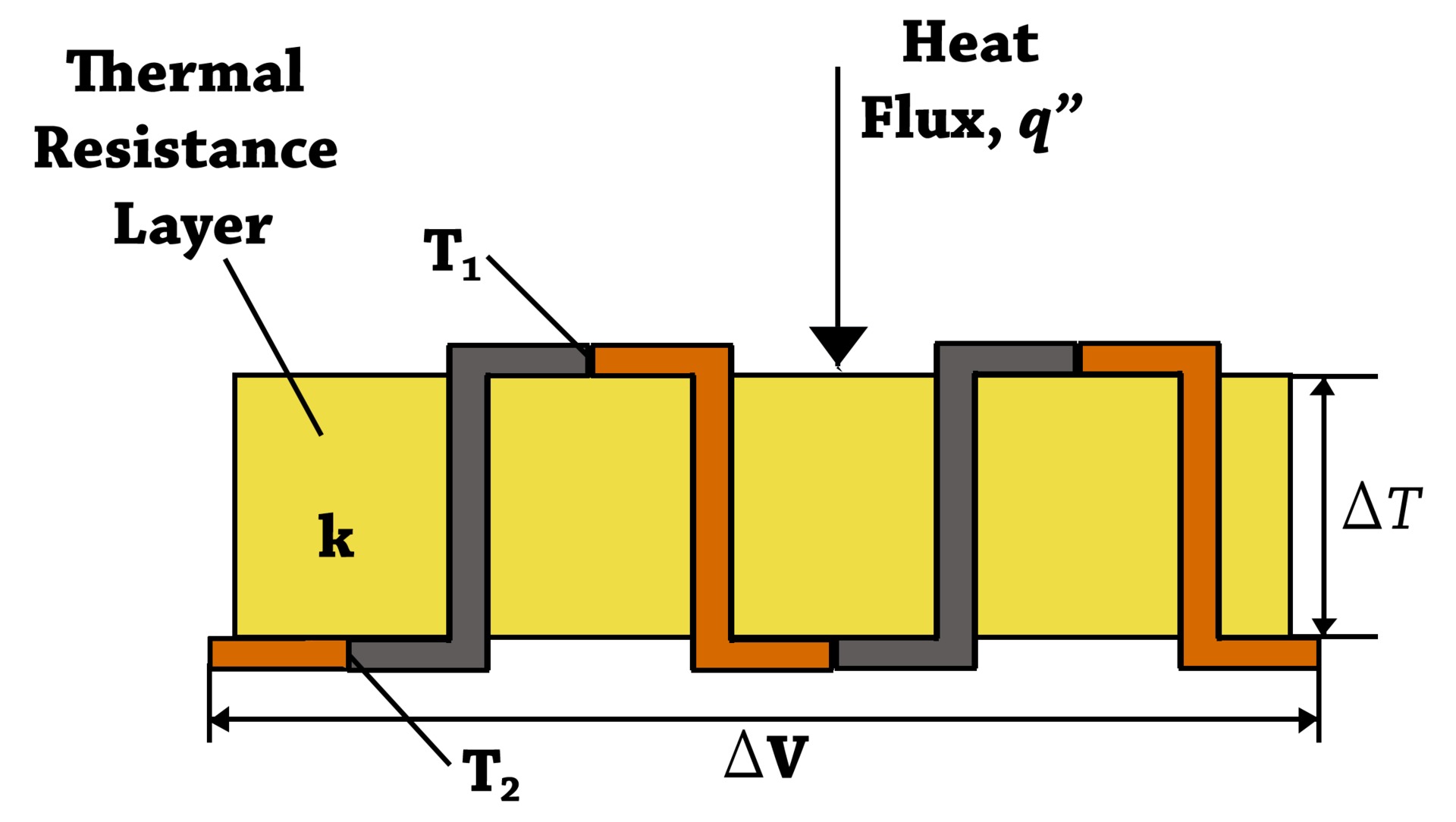
This is called a thermopile; when the heat source is radioactive material, as here, the device is called a radioisotope thermoelectric generator (RTG). The illustration shows the thermocouples strung out in a long line, but in an actual RTG you put the plutonium in a capsule and put the thermocouples in the wall of the capsule, with the outside joints attached to heat sinks. The plutonium heats up the inside joints to generate the current.
RTGs are more commonly used to power spacecraft, but there are a few dozen people still in the U.S. with plutonium-powered thermopile batteries in their pacemakers.
In pacemakers, the plutonium was sealed inside a titanium capsule, which was strong enough to survive an accident (such as a bullet impact or auto collision) or cremation. But Wikipedia says the technique was abandoned because of worries that the capsule wouldn't be absolutely certain to survive a cremation. (Cremation temperatures go up to around 1000°C; titanium melts at 1668°C.) Loose plutonium in the environment would be Very Bad.
(I wondered if there wasn't also worry about plutonium being recovered for weapons use, but the risk seems much smaller: you need several kilograms of plutonium to make a bomb, and a pacemaker has only around 135 mg, if I did the conversion from curies correctly. Even so, if I were in charge of keeping plutonium out of the wrong hands, I would still worry about this. It does not seem totally out of the realm of possibility that someone could collect 25,000 pacemakers. Opening 25,000 titanium capsules does sound rather tedious.)
Earlier a completely different nuclear-powered pacemaker was tried, based on promethium-powered betavoltaics. This is not a heat-conversion process. Instead, a semiconductor does some quantum physics magic with the electrons produced by radioactive beta decay. This was first demonstrated by Henry Moseley in 1913. Moseley is better-known for discovering that atoms have an atomic number, thus explaining the periodic table. The periodic table had previously been formulated in terms of atomic mass, which put some of the elements in the wrong order. Scientists guessed they were in the wrong order, because the periodicity didn't work, but they weren't sure why. Moseley was able to compute the electric charge of the atomic nucleus from spectrographic observations. I have often wondered what else Moseley would have done if he had not been killed in the European war at the age of 27.
It took a while to gather the information about this. Several of Wikipedia's articles on the topic are not up to their usual standards. The one about the radioisotope thermoelectric generator is excellent, though.
Thermopile illustration is by FluxTeq (Own work) CC BY-SA 4.0, via Wikimedia Commons.
[ Addendum 20180115: Commenters on Hacker News have pointed out that my concern about the use of plutonium in fission weapons is easily satisfied: the fuel in the batteries is Pu-238, which is not fissile. The plutonium to use in bombs is Pu-241, and indeed, when building a plutonium bomb you need to remove as much Pu-238 as possible, to prevent its non-fissile nuclei from interfering with the chain reaction. Interestingly, you can tell this from looking at the numbers: atomic nuclei with an odd number of neutrons are much more fissile than those with an even number. Plutonium is atomic number 94, so Pu-238 has an even number of neutrons and is not usable. The other isotope commonly used in fission is U-235, with 143 neutrons. I had planned to publish a long article today detailing the difficulties of gathering enough plutonium from pacemakers to make a bomb, but now I think I might have to rewrite it as a comedy. ]
[ Addendum 20170116: I published it anyway, with some editing. ]
[Other articles in category /tech] permanent link


